
In addition, the possibility of autophagy as a therapeutic target for cardiac hypertrophy will also be highlighted. In this review, we focus on how autophagy is regulated and what are the effects of altered autophagy on cardiac hypertrophy. 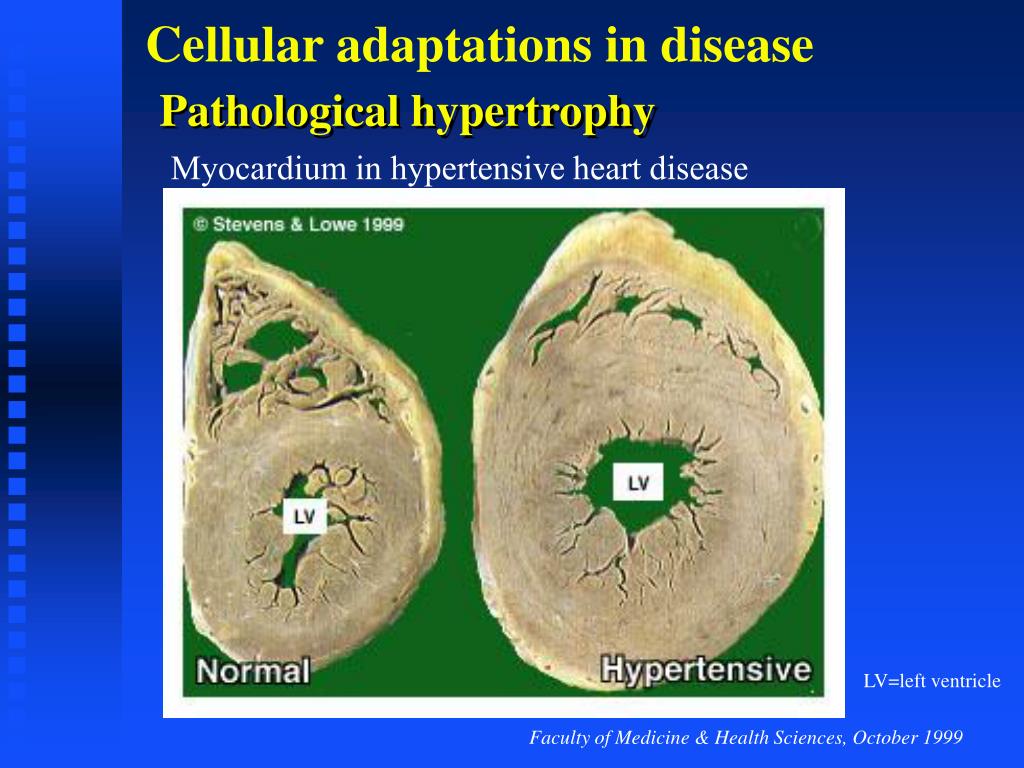
Recently, a number of studies have begun to show that autophagy plays important roles in cardiac hypertrophy under various pathological stimuli, thus indicating a possible link between autophagy dysregulation and cardiac hypertrophy. Dysregulation of autophagy is associated with many clinical disorders, such as cancer, neurodegeneration, infection and metabolic syndromes. Particularly, studies by using autophagy-defective animal models have elucidated the significant physiopathological functions of autophagy at whole-organism or tissue-specific levels. Constitutive autophagy is required for cell survival, while excessive autophagy triggers autophagic cell death, indicating dual roles of autophagy in pro- or anti-survival potential. Not only does autophagy play essential roles in tissue homeostasis by degradating long-lived proteins and unwanted organelles, it also acts as a recycling system to produce new energy and materials for the renovation of cells. Autophagy occurs in response to various stimuli, such as nutrient limitation and cellular stress. Further understanding the mechanisms underlying pathological cardiac hypertrophy would provide novel therapeutic strategies for the treatment of heart failure.Īutophagy is a conserved catabolic process by which cellular components are transported to and degraded in the lysosomes. The mechanisms of pathological cardiac hypertrophy have been studied at multiple levels, such as genetic mutations in structural proteins, alterations of molecular signaling pathways, detrimental changes in subcellular organelles and communications among various cell types in hearts. Although pathological cardiac hypertrophy initially compensates for the biomechanical and physiopathological stimuli, it progresses generally to detrimental cardiac remodeling and heart failure, which is a clinical risk factor for death. At cellular level, pathological cardiac hypertrophy is characterized by an increase in cardiomyocyte size, disarrangement of sarcomeric structure, enhanced protein synthesis and re-expression of fetal genes. aging, myocardial ischemia and hypertension. Pathological cardiac hypertrophy refers to the enlargement of heart in a variety of diseases e.g. Keywords: autophagy, cardiac hypertrophy, heart failure, therapeutic target Introduction Moreover, the therapeutic strategies targeting autophagy for cardiac hypertrophy will also be discussed. Here we review the recent progresses concerning the functions of autophagy in cardiac hypertrophy induced by various hypertrophic stimuli. Sophisticatedly regulated autophagy protects heart from various physiological and pathological stimuli by degradating and recycling of protein aggregates, lipid drops, or organelles. Accumulating evidence has revealed a tight link between cardiomyocyte autophagy and cardiac hypertrophy. Autophagy is a dynamic self-degradative process necessary for the maintenance of cellular homeostasis. However, a long-term exposure to the stress makes heart progress to heart failure. Pathological cardiac hypertrophy is the response of heart to various biomechanical and physiopathological stimuli, such as aging, myocardial ischemia and hypertension. Select the file that you have just downloaded and select import option Reference Manager (RIS). Functions of Autophagy in Pathological Cardiac Hypertrophy.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
